202603 | Nearly Half of Implant Restoration Kits Are European, Small and Medium-Sized Implant Brands Successively Approved
 |
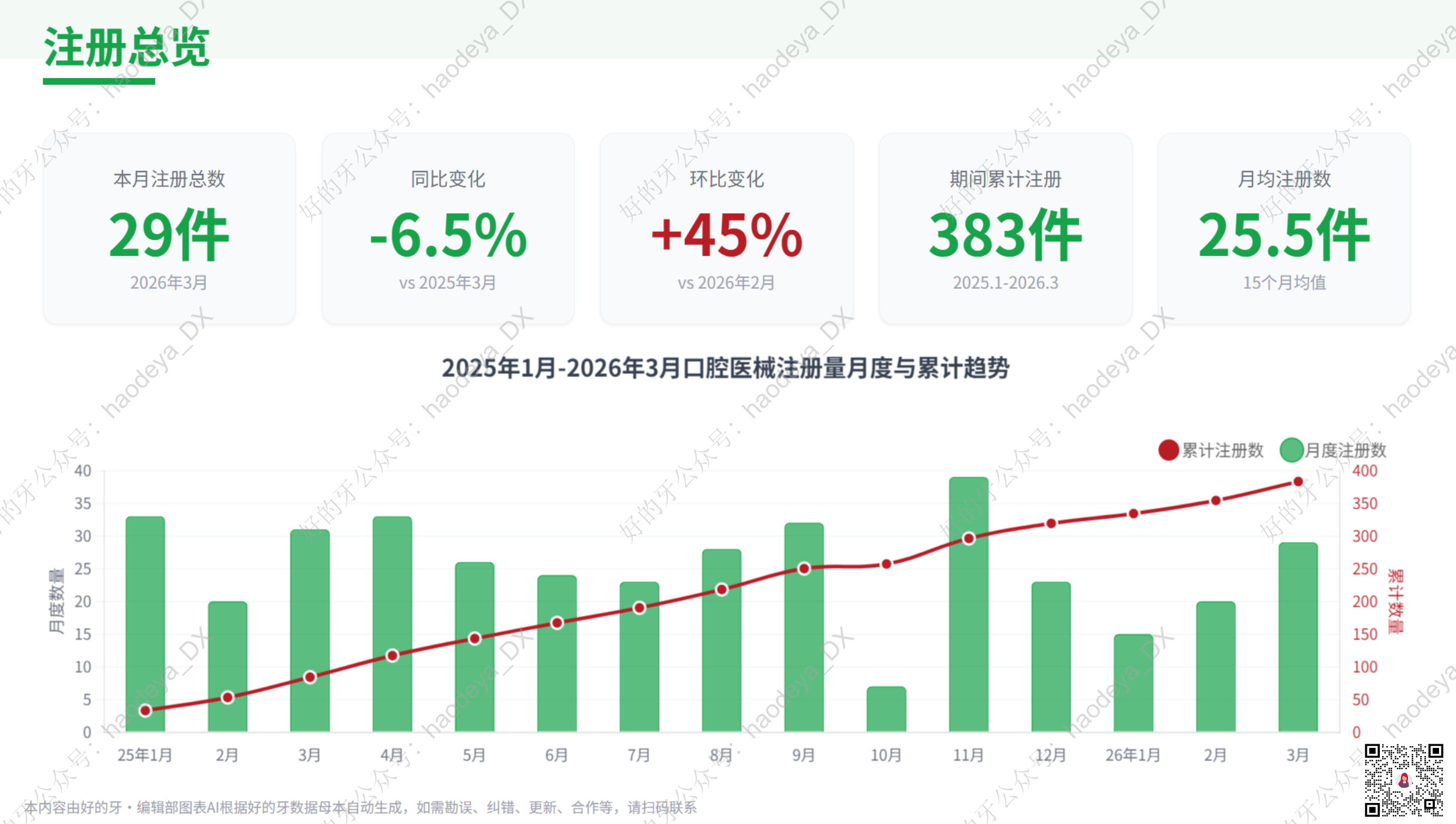
In March 2026, the registration landscape for dental medical devices revealed significant shifts, highlighting both challenges and opportunities within the industry. The total number of registrations saw a notable decrease from the previous year, yet a month-on-month rebound indicates a recovery trend.Among the registered products, a substantial portion were domestic, reflecting a growing capacity for local manufacturing. However, the import scene tells a different story, with a shift in focus from prosthetic accessories to implant systems, as small-to-medium-sized European brands gain approval. This change underscores the evolving dynamics of clinical demand and the competitive landscape.As the industry grapples with these transitions, manufacturers are urged to adapt strategically to maintain relevance. The ongoing developments in product registration not only shed light on market trends but also signal a crucial phase for innovation and collaboration in dental technology…
This content is available to DentalGoodNews Pro members. Subscribe to read full article

| About DGN:DentalGoodNews (DGN) is a trusted professional media platform dedicated to the global dental industry. We deliver in-depth coverage of corporate news, policy & regulation, investment & funding, and clinical frontiers — serving dental institutions, device manufacturers, investors, and industry researchers worldwide. Contact us: haodeya@dongxizixun.com |
- 🧠 Real-time notifications for policy/data updates
- 📚 Access to selected industry resource packs
- 🧾 Membership update reminders + unlock notifications
- 🎉 Community events & coupon benefits
- 💬 Online customer service Q&A (content/download/inquiry)
