AI automatically detects dental caries and gingival recession, 3Shape diagnostic software receives FDA clearance [SOURCE] AI automatically detects dental caries and gingival recession, 3Shap…
 |
| Source: 3Shape official website |
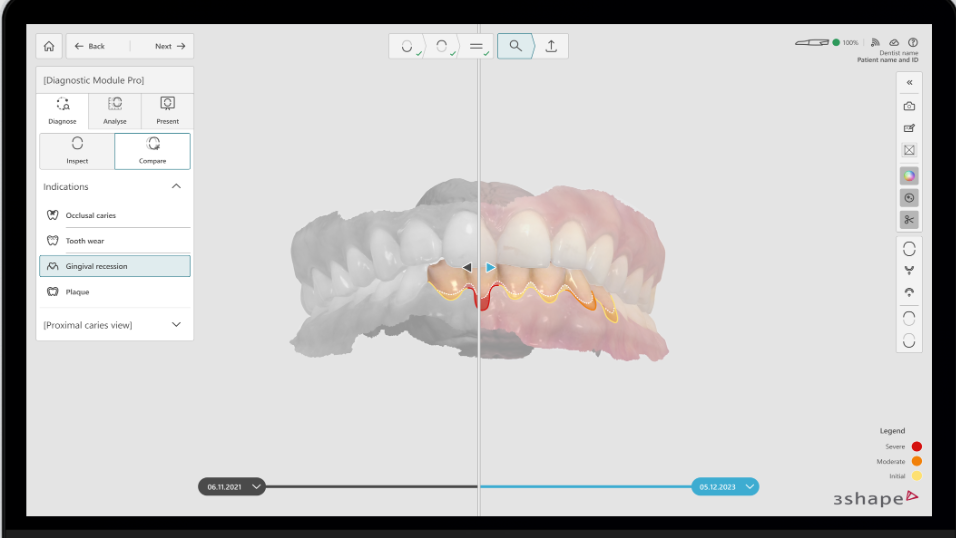
DentalGoodNews|April 28, 2026 - 3Shape announced that its Dx diagnostic software (including Dx Plus and Dx Standard) has received 510(k) clearance from the U.S. Food and Drug Administration (FDA). This clearance allows the Dx (R1) software to be used by qualified dental professionals in U.S. dental clinics for oral diagnosis and assessment of tooth and gingival changes in adult patients.
As a core component of 3Shape's digital ecosystem, the cleared Dx software offers two levels of diagnostic support. Dx Plus is the AI-assisted diagnostic version, capable of detecting key conditions such as dental caries (occlusal and proximal surfaces), dental plaque, tooth wear, and gingival recession. Dx Standard focuses on helping dentists manually track and quantify changes in patients' teeth. In terms of device compatibility, Dx Plus is specifically developed for the latest TRIOS 6 intraoral scanner, while Dx Standard supports all TRIOS models except TRIOS Core.
3Shape CEO Jacob Paulsen stated in the announcement that the Dx software transforms clinical findings into intuitive visual results, providing doctors with a new, efficient way to communicate with patients and enhancing patient confidence in treatment recommendations. According to a previous report by DENTALGOODNEWS (Leading Dental Industry Media, DGN), 3Shape launched the TRIOS 6 and its accompanying AI diagnostic software at the 2025 International Dental Show (IDS), emphasizing improved clinic operational efficiency through an integrated workflow.
In terms of international expansion, the Dx software has been sold and used clinically in multiple countries and regions, including Europe, the Middle East, Africa, South America, and Canada, since 2025. As of press time, 3Shape has not disclosed the suggested retail price or launch date for this product in the Chinese market.
| About DGN:DentalGoodNews (DGN) is a trusted professional media platform dedicated to the global dental industry. We deliver in-depth coverage of corporate news, policy & regulation, investment & funding, and clinical frontiers — serving dental institutions, device manufacturers, investors, and industry researchers worldwide. Contact us: haodeya@dongxizixun.com |
- 🧠 Real-time notifications for policy/data updates
- 📚 Access to selected industry resource packs
- 🧾 Membership update reminders + unlock notifications
- 🎉 Community events & coupon benefits
- 💬 Online customer service Q&A (content/download/inquiry)
